Cells are able to deform, migrate or integrate external mechanical cues such as forces or rigidity of the environment. These capabilities are essential all along the life of an organism starting from development to pathologies.
Our team developed during the last years a new approach to access to these properties at the scale of in vitro networks, subcellular structures in vitro or in cellulo, isolated cells and cell monolayer. We use dipolar forces that develop in superparamagnetic colloids in an homogenous magnetic field to follow the deformation of biological structures under controlled forces (applied and tuned by the intensity of the magnetic field). In addition, the self-assembly of the colloids into chains allow high throughput of the measurements to be obtained.
 Based on this technique we have studied the link between architecture and mechanics of dense branched networks resembling the ones found in the leading edge of a moving cell (Arp2/3 machinery) using a minimal mix of proteins (Pujol et al. 2012, Tavacoli et al 2013, Bauër et al. 2017) or yeast extract (Planade et al. 2019, Belbahri et al 2022)
Based on this technique we have studied the link between architecture and mechanics of dense branched networks resembling the ones found in the leading edge of a moving cell (Arp2/3 machinery) using a minimal mix of proteins (Pujol et al. 2012, Tavacoli et al 2013, Bauër et al. 2017) or yeast extract (Planade et al. 2019, Belbahri et al 2022)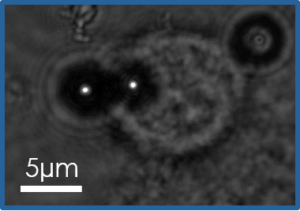
More recently we have implemented the technique inside live cells to assess the biophysics of the cell cortex (Laplaud et al 2021)
This work is a daily collaboration with Julien Heuvingh (PMMH, ESPCI).
External Collaborations:
- Guillaume Romet-Lemonne & Antoine Jégou (IJM, Paris)
- Alphée Michelot (IBDM, Marseille, France)
- Matthieu Piel (IPGG, Institut Curie, Paris, France)
- Nicolas Borghi (IJM, Paris)
- Martin Lenz (LPTMS, Orsay, France)
Fundings:
Project FruSA granted by the French ANR ANR-22-CE30-0024
Project CoMePol granted by the French ANR ANR-21-CE13-0048
Project MUSCACTIN granted by the French ANR 15-CE13-0004, 2015-2020.
Previous PhD Students and Post-Doc:
Thomas Pujol (PhD, 2009-2012), Joseph Tavacoli (Post-Doc, 2011-2013), Pierre Bauër (PhD, 2012-2015), Jessica Planade (PhD, 2013-2016), Cesar Vanecia-Gallardo (2017-2019), Reda Belbahri (2016-2019), Magdalean Kopec (2019-2023).
 Home
Home